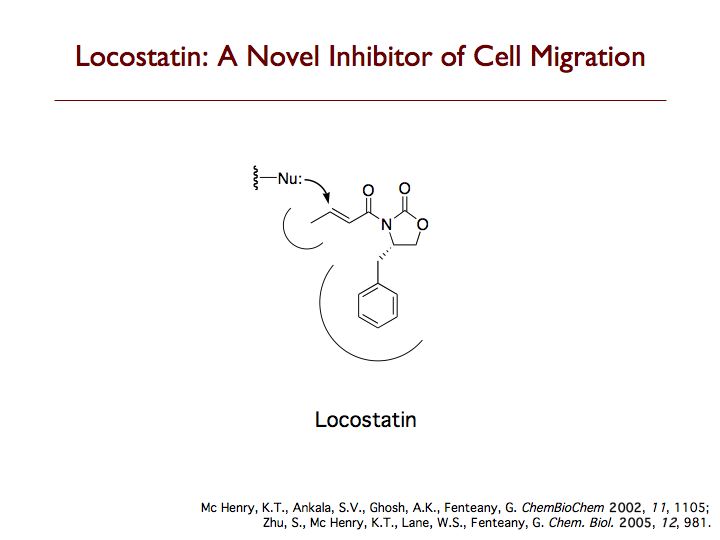
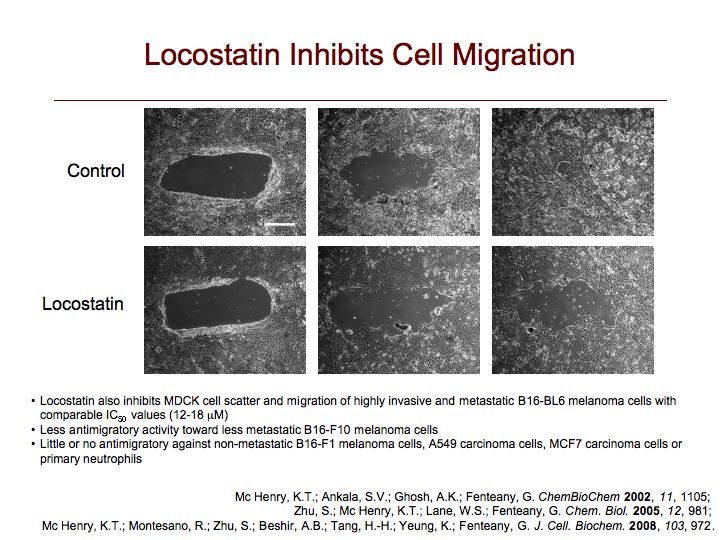
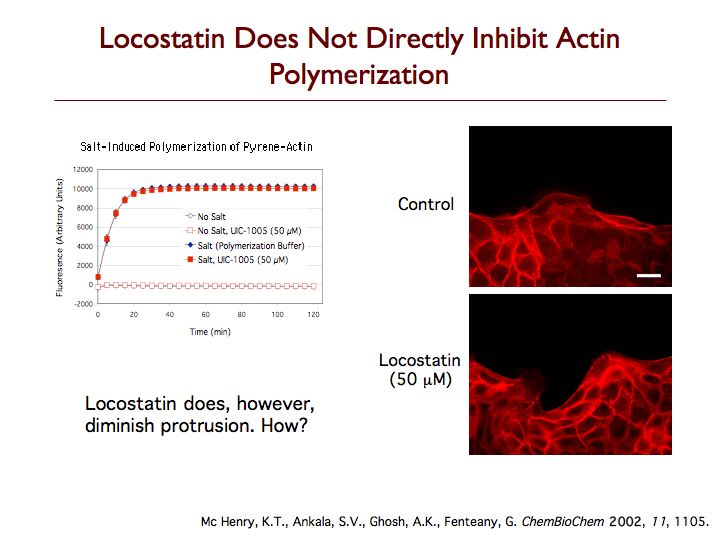
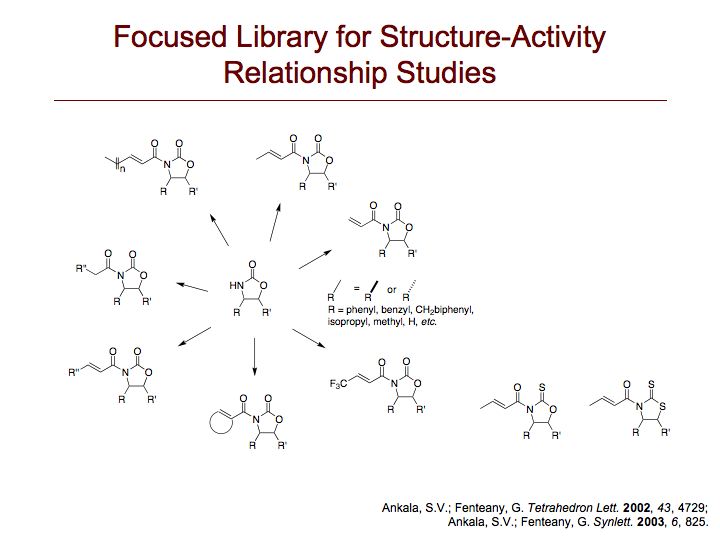
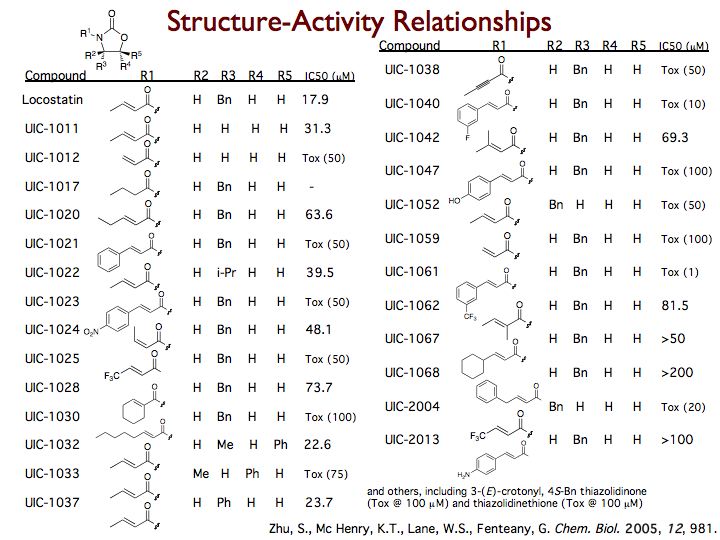
Locostatin
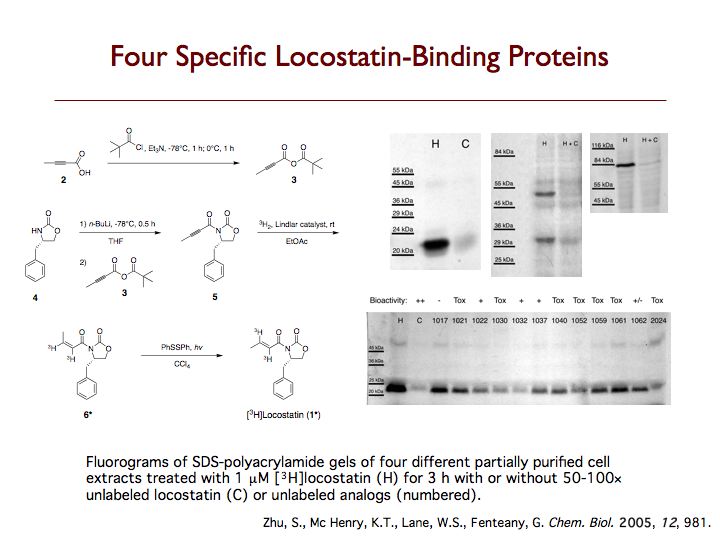
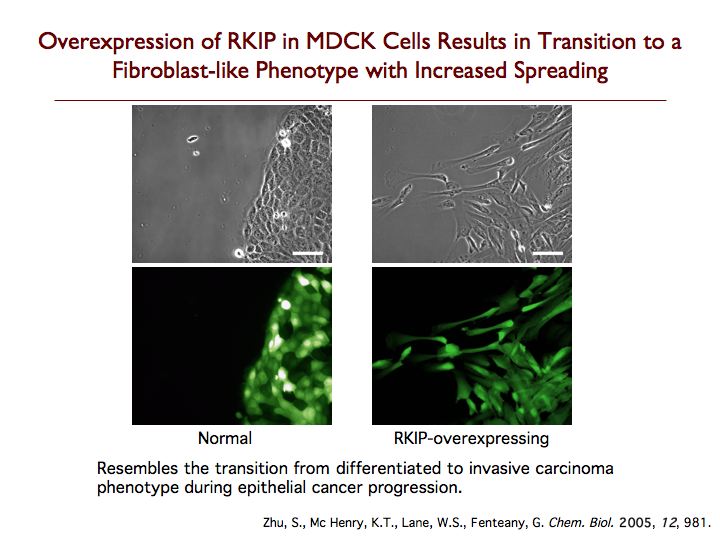
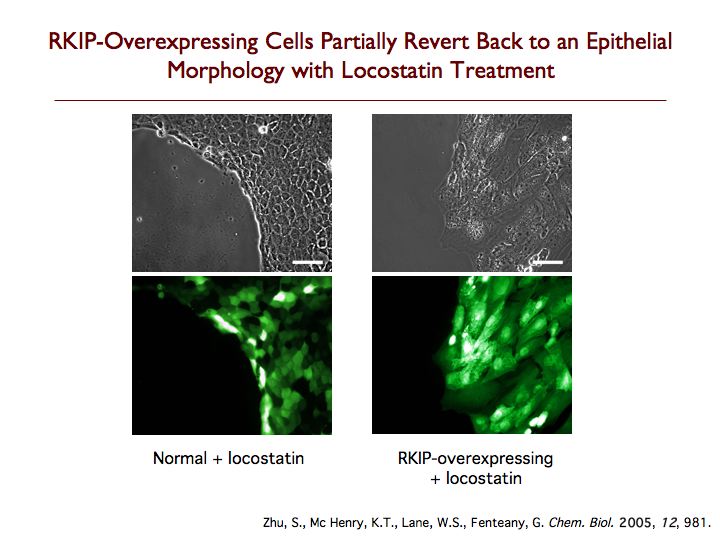
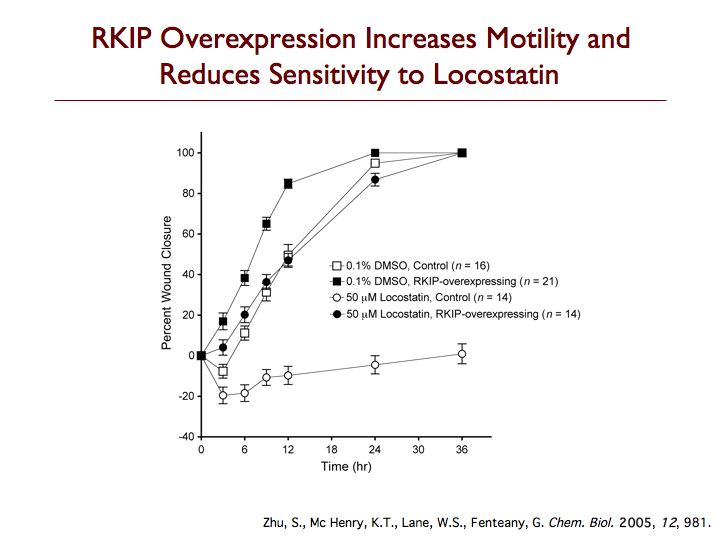
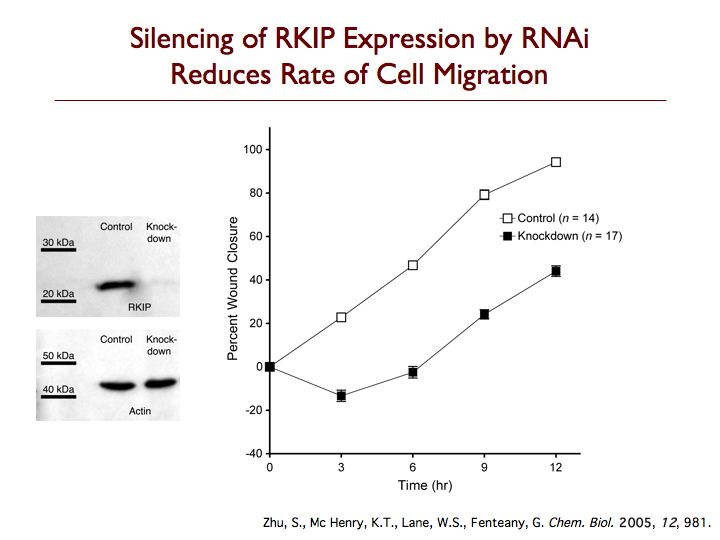
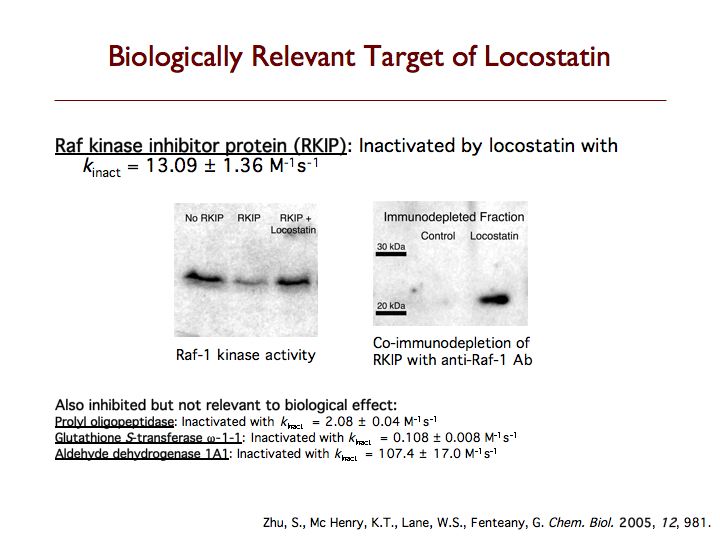
Raf kinase inhibitor protein (RKIP) is a modulator of cell signaling that functions as an endogenous inhibitor of multiple kinases. Here we demonstrate a new positive role for RKIP in the regulation of cell locomotion. We discovered that RKIP is the relevant cellular target of locostatin, a novel cell migration inhibitor. Locostatin abrogates RKIP's ability to bind and inhibit Raf-1 kinase, making it the first known inhibitor of RKIP, one that acts by disrupting a protein-protein interaction, an uncommon mode of action for a small molecule. Small interfering RNA-mediated silencing of RKIP expression also reduces cell migration rate. Overexpression of RKIP converts epithelial cells to a highly migratory fibroblast-like phenotype, with dramatic reduction in the sensitivity of cells to locostatin. RKIP is therefore a target of the compound and functions as a regulator of cell motility.
Exploring the role of RKIP in cell adhesion, we found that overexpression of RKIP in epithelial cells increases adhesion to the substratum, while decreasing adhesion of the cells to one another. The level of the adherens junction protein E-cadherin declines profoundly, and there is loss of normal localization of the tight junction protein ZO-1, while expression of the cell-substratum adhesion protein β1 integrin dramatically increases. The cells also display increased adhesion and spreading on multiple substrata, including collagen, gelatin, fibronectin and laminin. In three-dimensional culture, RKIP overexpression leads to marked cell elongation and extension of long membrane protrusions into the surrounding matrix, and the cells do not form hollow cysts. RKIP-overexpressing cells generate considerably more contractile traction force than do control cells. In contrast, RNA interference-based silencing of RKIP expression results in decreased cell-substratum adhesion in both MDCK and MCF7 human breast adenocarcinoma cells. Treatment of MDCK and MCF7 cells with locostatin, a direct inhibitor of RKIP and cell migration, also reduces cell-substratum adhesion. Silencing of RKIP expression in MCF7 cells leads to a reduction in the rate of wound closure in a scratch-wound assay, although not as pronounced as that previously reported for RKIP-knockdown MDCK cells. These results suggest that RKIP has important roles in the regulation of cell adhesion, positively controlling cell-substratum adhesion while negatively controlling cell-cell adhesion, and underscore the complex functions of RKIP in cell physiology.
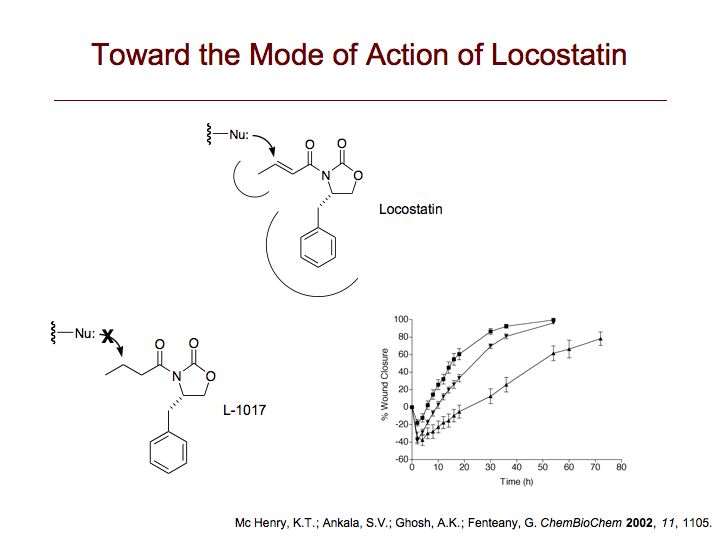
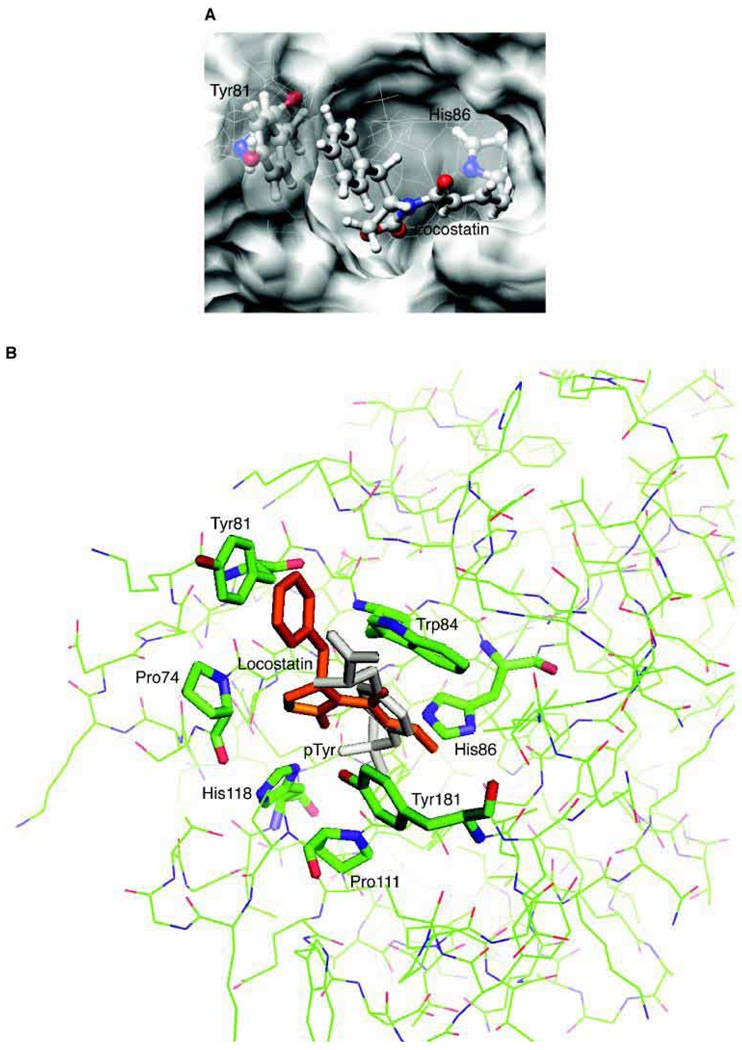
RKIP interacts with a number of different proteins and regulates multiple signaling pathways. We have found that locostatin not only disrupts interactions of RKIP with Raf-1 kinase, but also with G protein-coupled receptor kinase 2. In contrast, we found that locostatin does not disrupt binding of RKIP to two other proteins: inhibitor of κB kinase α and transforming growth factor β-activated kinase 1. These results thus imply that different proteins interact with different regions of RKIP. Locostatin's mechanism of action involves modification of a nucleophilic residue on RKIP. We observed that after binding RKIP, part of locostatin is slowly hydrolyzed, leaving a smaller RKIP-butyrate adduct. We identified the residue alkylated by locostatin as His86, a highly conserved residue in RKIP's ligand-binding pocket. Computational modeling of the binding of locostatin to RKIP suggested that the recognition interaction between small molecule and protein ensures that locostatin's electrophilic site is poised to react with His86. Furthermore, binding of locostatin would sterically hinder binding of other ligands in the pocket. These data provide a basis for understanding how locostatin disrupts particular interactions of RKIP with RKIP-binding proteins and demonstrate its utility as a probe of specific RKIP interactions and functions.
More on the mechanism of cell sheet migration.
More on signaling pathways to cell motility.